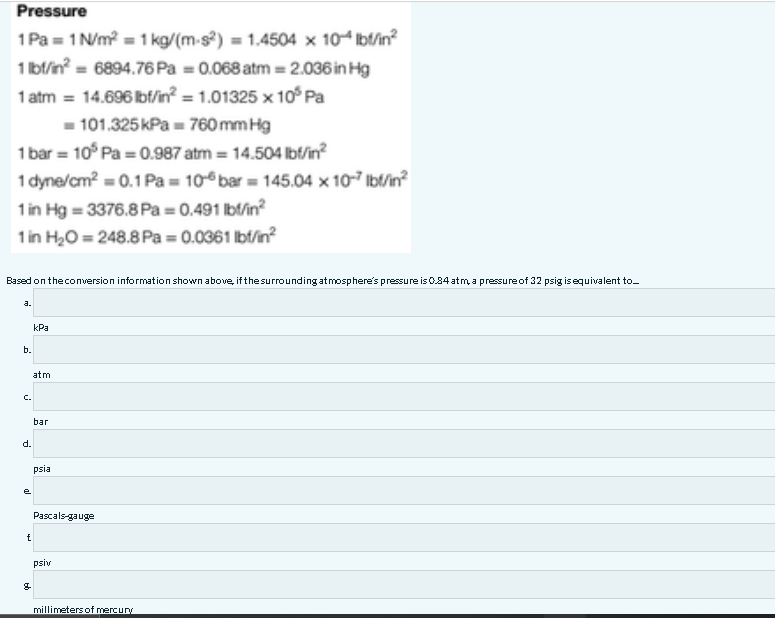
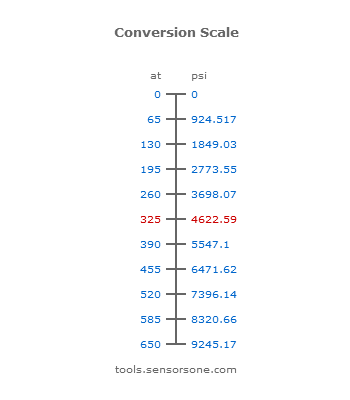
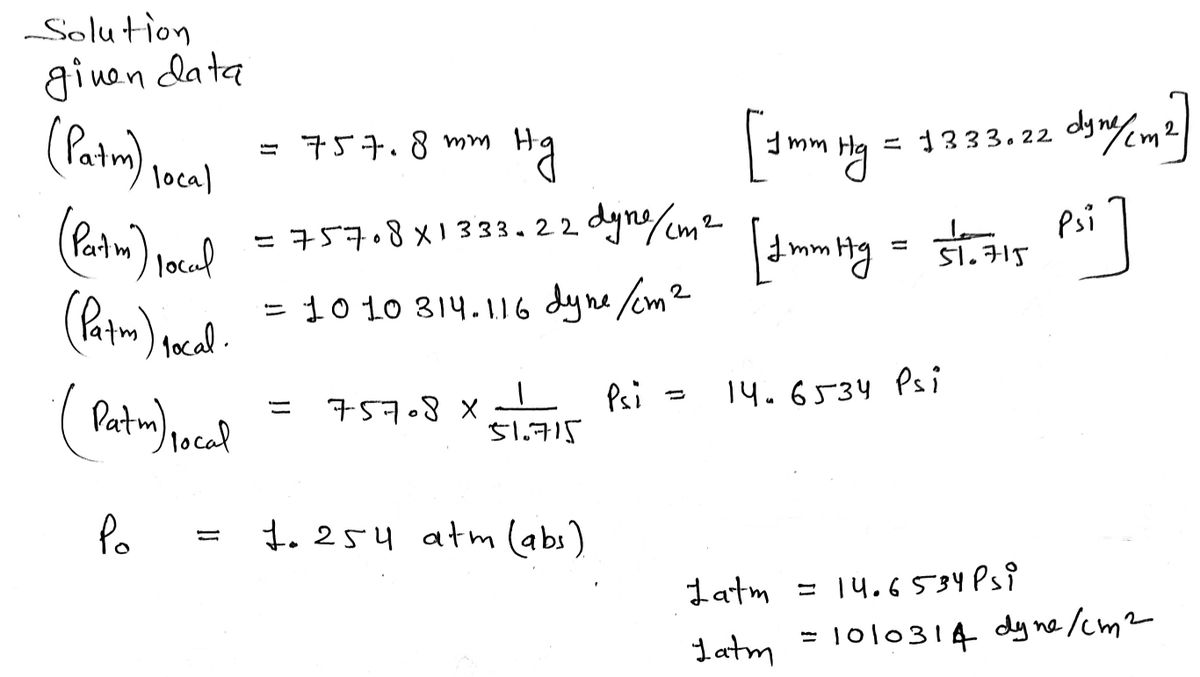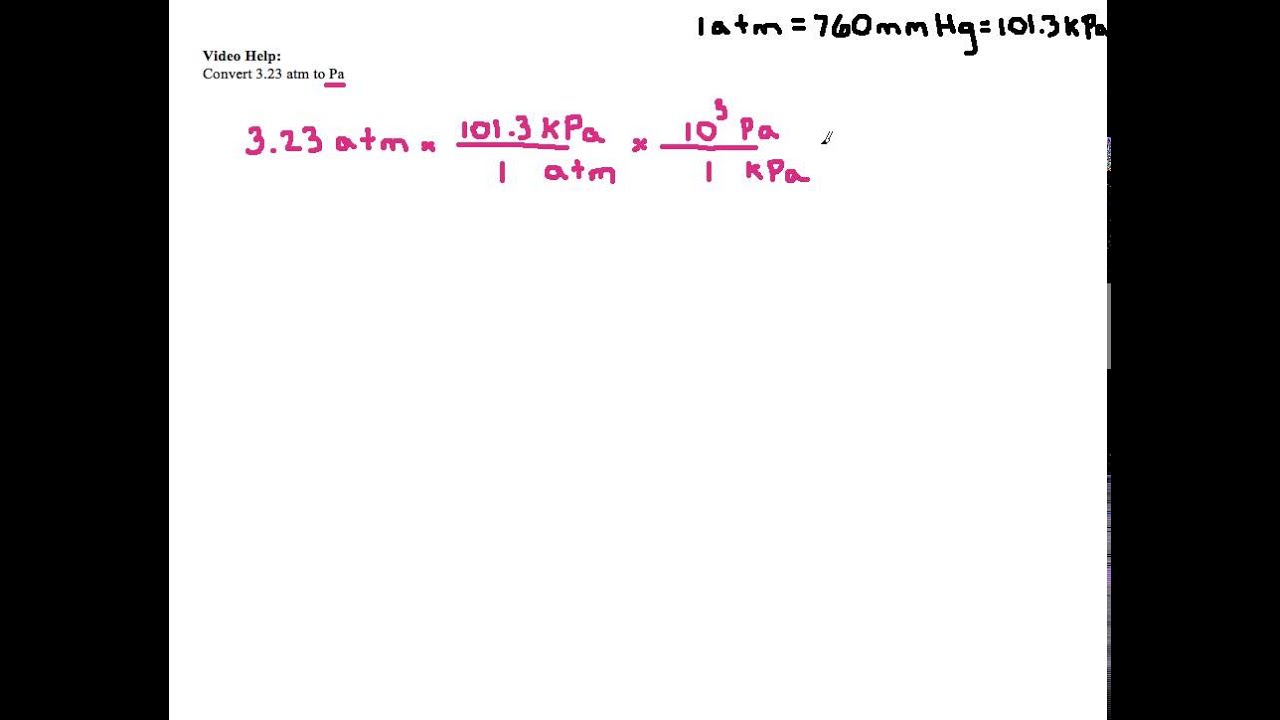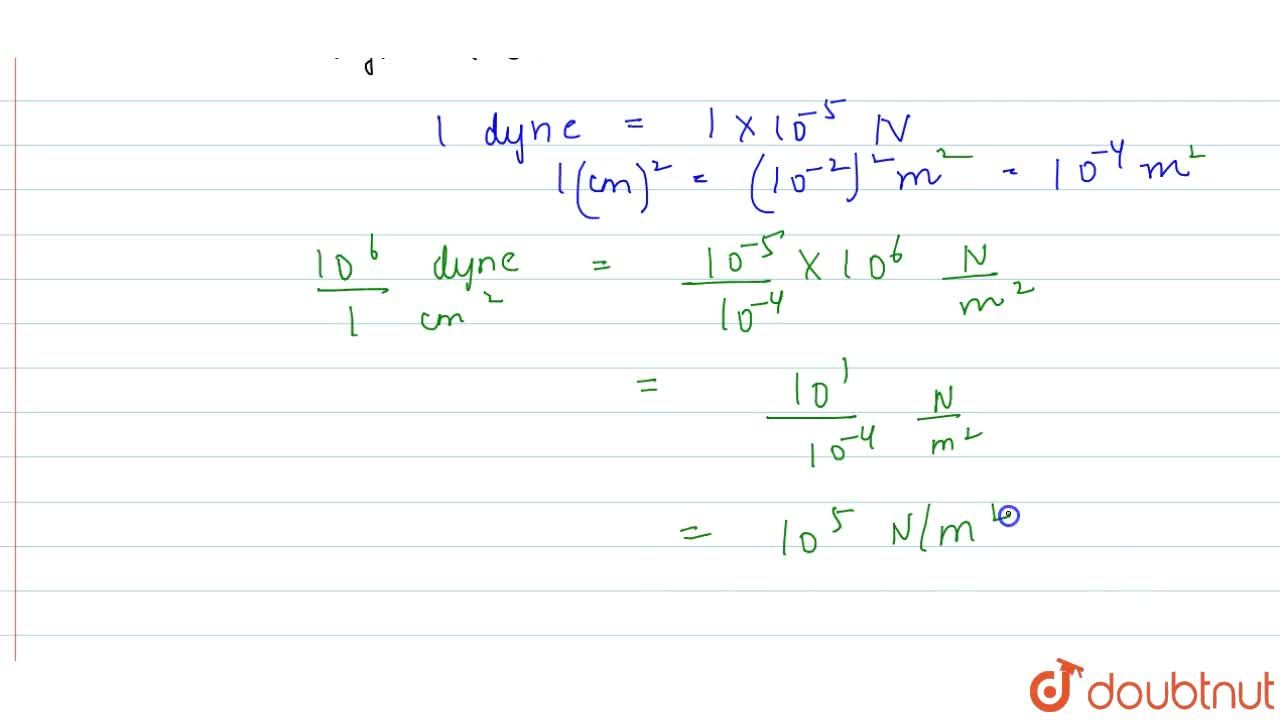Vacuum increases the mean-free-path of gas molecules.mean-free-path Vacuum prevents chemical reaction. Vacuum removes contaminants from surfaces.contaminants. - ppt download

If the value of atmospheric pressure is `10^(6)` dyne `cm^(-2)`, find its value in SI units. - YouTube
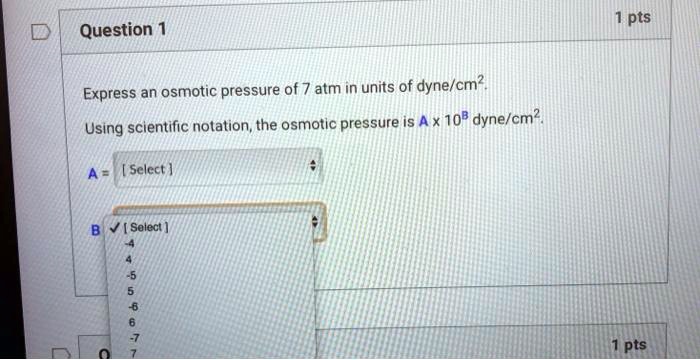
SOLVED: pts Question 1 Express an osmotic pressure of 7 atm in units of ' dyne/cm?. Using scientific nolation, the osmotic pressure is A x 108 dyne/cm?, Sclect | Solect pts

What will be the pressure in dyne cm^-2 , due to a water column of height 12.5cm ? ( g = 980cm s^-2 ) (density of water 10^3kg m^-3 ).